Vanadium tetrachloride
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Vanadium tetrachloride
Vanadium(IV) chloride | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.028.692 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| VCl4 | |||
| Molar mass | 192.75 g/mol | ||
| Appearance | bright red liquid, moisture sensitive | ||
| Odor | pungent | ||
| Density | 1.816 g/cm3, liquid | ||
| Melting point | −24.5 °C (−12.1 °F; 248.7 K) | ||
| Boiling point | 148 °C (298 °F; 421 K) | ||
| decomposes | |||
| Solubility | soluble in CH2Cl2 | ||
| Vapor pressure | 7.9 Pa | ||
| +1130.0·10−6 cm3/mol | |||
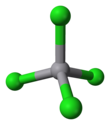
| Structure | |||
| tetrahedral | |||
| 0 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
toxic; oxidizer; hydrolyzes to release HCl | ||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
160 mg/kg (rat, oral) | ||
| Related compounds | |||
Other anions
|
vanadium tetrafluoride, vanadium disulfide, vanadium tetrabromide | ||
Other cations
|
titanium tetrachloride, chromium tetrachloride, niobium tetrachloride, tantalum tetrachloride | ||
Related compounds
|
vanadium trichloride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Vanadium tetrachloride is the inorganic compound with the formula VCl4. This reddish-brown liquid serves as a useful reagent for the preparation of other vanadium compounds.
Synthesis, bonding, basic properties
[edit]With one more valence electron than diamagnetic TiCl4, VCl4 is a paramagnetic liquid. It is one of only a few paramagnetic compounds that is liquid at room temperature.
VCl4 is prepared by chlorination of vanadium metal. VCl5 does not form in this reaction; Cl2 lacks the oxidizing power to attack VCl4. VCl5 can however be prepared indirectly from VF5 at −78 °C.[1]
Reactions
[edit]Consistent with its high oxidizing power, VCl4 reacts with HBr at -50 °C to produce VBr3. The reaction proceeds via VBr4, which releases Br2 during warming to room temperature.[2]
- 2 VCl4 + 8 HBr → 2 VBr3 + 8 HCl + Br2
VCl4 forms adducts with many donor ligands, for example, VCl4(THF)2.
It is the precursor to vanadocene dichloride.
Organic chemistry
[edit]In organic synthesis, VCl4 is used for the oxidative coupling of phenols. For example, it converts phenol into a mixture of 4,4'-, 2,4'-, and 2,2'-biphenols:[3]
- 2 C6H5OH + 2 VCl4 → HOC6H4–C6H4OH + 2 VCl3 + 2 HCl
Applications
[edit]VCl4 is a catalyst for the polymerization of alkenes, especially those useful in the rubber industry. The underlying technology is related to Ziegler–Natta catalysis, which involves the intermediacy of vanadium alkyls.
Safety considerations
[edit]VCl4 is a volatile, aggressive oxidant that readily hydrolyzes to release HCl.
References
[edit]- ^ Tamadon, Farhad; Seppelt, Konrad (2013). "The Elusive Halides VCl5, MoCl6, and ReCl6". Angew. Chem. Int. Ed. 52 (2): 767–769. doi:10.1002/anie.201207552. PMID 23172658.
- ^ Calderazzo, F.; Maichle-Mössmer, C.; G., Pampaloni; J., Strähle (1993). "Low-temperature Syntheses of Vanadium(III) and Molybdenum(IV) Bromides by Halide Exchange". Dalton Transactions (5): 655–8. doi:10.1039/DT9930000655.
- ^ O’Brien, M. K.; Vanasse, B. (2001). "Vanadium(IV) Chloride". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York, NY: J. Wiley & Sons. doi:10.1002/047084289X.rv001. ISBN 0471936235.