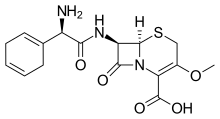
Cefroxadine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | ~1 hour |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.052.157 |
| Chemical and physical data | |
| Formula | C16H19N3O5S |
| Molar mass | 365.40 g·mol−1 |
| 3D model (JSmol) | |
| |
Cefroxadine (INN, trade names Oraspor and Cefthan-DS) is a cephalosporin antibiotic. It is structurally related to cefalexin, and both drugs share a similar spectrum of activity.[1]
It is available in Italy.[2]
Synthesis
[edit]Cefroxadine can be prepared by several routes, including one in which the enol is methylated with diazomethane as a key step. A rather more involved route starts with comparatively readily available phenoxymethylpenicillin sulfoxide benzhydryl ester (1).

This undergoes fragmentation when treated with benzothiazole-2-thiol to give 2. Ozonolysis (reductive work-up) cleaves the olefinic linkage and the unsymmetrical disulfide moiety is converted to a tosyl thioester (3). The enol moiety is methylated with diazomethane, the six-membered ring is closed by reaction with 1,5-diazabicyclo[5.4.0]undec-5-ene (DBU), and the ester protection is removed with trifluoroacetic acid to give 4. The amide side chain is removed by the usual PCl5/dimethylaniline sequence followed by reamidation with the appropriate acid chloride to give cefroxadine (5).
See also
[edit]References
[edit]- ^ Yasuda K, Kurashige S, Mitsuhashi S (July 1980). "Cefroxadine (CGP-9000), an orally active cephalosporin". Antimicrobial Agents and Chemotherapy. 18 (1): 105–10. doi:10.1128/AAC.18.1.105. PMC 283947. PMID 6998373.
- ^ [No authors listed]. "Oraspor". Prontuario.it (in Italian). Elsevier. Retrieved 2010-07-31.
- ^ DE 2331133, Bickel, Hans & Scartazzini, Riccardo, "Enolderivate [Enol derivatives]", published 1974-01-17, assigned to Ciba-Geigy AG
- ^ R. Scartazzini, H. Bickel, U.S. patent 4,073,902 (1978 to Ciba-Geigy).
- ^ R. B. Woodward and H. Bickel, U.S. patent 4,147,864 (1979); Chem. Abstr., 91, 74633J (1979).
