Metal cluster compound
Metal cluster compounds are a molecular ion or neutral compound composed of three or more metals and featuring significant metal-metal interactions.[2]
Transition metal carbonyl clusters
[edit]The development of metal carbonyl clusters such as Ni(CO)4 and Fe(CO)5 led quickly to the isolation of Fe2(CO)9 and Fe3(CO)12. Rundle and Dahl discovered that Mn2(CO)10 featured an "unsupported" Mn-Mn bond, thereby verifying the ability of metals to bond to one another in molecules. In the 1970s, Paolo Chini demonstrated that very large clusters could be prepared from the platinum metals, one example being [Rh13(CO)24H3]2−. This area of cluster chemistry has benefited from single-crystal X-ray diffraction.

Many metal carbonyl clusters contain ligands aside from CO. For example, the CO ligand can be replaced with myriad alternatives such as phosphines, isocyanides, alkenes, hydride, etc. Some carbonyl clusters contain two or more metals. Others contain carbon vertices. One example is the methylidyne-tricobalt cluster [Co3(CH)(CO)9].[3] The above-mentioned cluster serves as an example of an overall zero-charged (neutral) cluster. In addition, cationic (positively charged) rather than neutral organometallic trimolybdenum[4][5] or tritungsten[6] clusters are also known. The first representative of these ionic organometallic clusters is [Mo3(CCH3)2(O2CCH3)6(H2O)3]2+.
Transition metal halide clusters
[edit]
The halides of low-valent early metals often are clusters with extensive M-M bonding. The situation contrasts with the higher halides of these metals and virtually all halides of the late transition metals, where metal-halide bonding is replete.
Transition metal halide clusters are prevalent for the heavier metals: Zr, Hf, Nb, Ta, Mo, W, and Re. For the earliest metals Zr and Hf, interstitial carbide ligands are also common. One example is Zr6CCl12.[7] One structure type features six terminal halides and 12 edge-bridging halides. This motif is exemplified by tungsten(III) chloride, [Ta6Cl18]4−,[8] Another common structure has six terminal halides and 8 bridging halides, e.g. Mo6Cl142−.
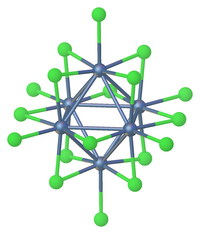
Many of the early metal clusters can only be prepared when they incorporate interstitial atoms.
In terms of history, Linus Pauling showed that "MoCl2" consisted of Mo6 octahedra. F. Albert Cotton established that "ReCl3" in fact features subunits of the cluster Re3Cl9, which could be converted to a host of adducts without breaking the Re-Re bonds. Because this compound is diamagnetic and not paramagnetic the rhenium bonds are double bonds and not single bonds. In the solid state further bridging occurs between neighbours and when this compound is dissolved in hydrochloric acid a Re3Cl123− complex forms. An example of a tetranuclear complex is hexadecamethoxytetratungsten W4(OCH3)12 with tungsten single bonds. A related group of clusters with the general formula MxMo6X8 such as PbMo6S8. These sulfido clusters are called Chevrel phases.
Fe-S clusters in biology
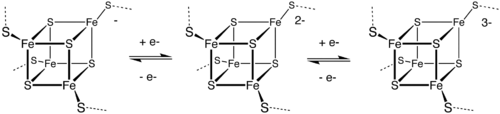
[edit]In the 1970s, ferredoxin was demonstrated to contain Fe4S4 clusters and later nitrogenase was shown to contain a distinctive MoFe7S9 active site.[10] The Fe-S clusters mainly serve as redox cofactors, but some have a catalytic function. In the area of bioinorganic chemistry, a variety of Fe-S clusters have also been identified that have CO as ligands.

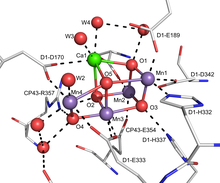
FeMoco, the active site of most nitrogenases, features a Fe7MoS9C cluster.[11]
Zintl clusters
[edit]Zintl compounds feature naked anionic clusters that are generated by reduction of heavy main group p elements, mostly metals or semimetals, with alkali metals, often as a solution in anhydrous liquid ammonia or ethylenediamine.[12] Examples of Zintl anions are [Bi3]3−, [Sn9]4−, [Pb9]4−, and [Sb7]3−.[13] Although these species are called "naked clusters," they are usually strongly associated with alkali metal cations. Some examples have been isolated using cryptate complexes of the alkali metal cation, e.g., [Pb10]2− anion, which features a capped square antiprismatic shape.[14] According to Wade's rules (2n+2) the number of cluster electrons is 22 and therefore a closo cluster. The compound is prepared from oxidation of K4Pb9 [15] by Au+ in PPh3AuCl (by reaction of tetrachloroauric acid and triphenylphosphine) in ethylene diamine with 2.2.2-crypt. This type of cluster was already known as is the endohedral Ni@Pb102− (the cage contains one nickel atom). The icosahedral tin cluster Sn122− or stannaspherene anion is another closed shell structure observed (but not isolated) with photoelectron spectroscopy.[16][17] With an internal diameter of 6.1 Ångstrom, it is of comparable size to fullerene and should be capable of containing small atoms in the same manner as endohedral fullerenes, and indeed exists a Sn12 cluster that contains an Ir atom: [Ir@Sn12]3−.[18]
Metalloid clusters
[edit]Elementoid clusters are ligand-stabilized clusters of metal compounds that possess more direct element-element than element-ligand contacts. Examples of structurally characterized clusters feature ligand stabilized cores of Al77, Ga84, and Pd145.[19]
Intermetalloid clusters
[edit]These clusters consist of at least two different (semi)metallic elements, and possess more direct metal-metal than metal-ligand contacts. The suffix "oid" designate that such clusters possess at a molecular scale, atom arrangements that appear in bulk intermetallic compounds with high coordination numbers of the atoms, such as for example in Laves phase and Hume-Rothery phases.[20] Ligand-free intermetalloid clusters include also endohedrally filled Zintl clusters.[13][21] A synonym for ligand-stabilized intermetalloid clusters is "molecular alloy". The clusters appear as discrete units in intermetallic compounds separated from each other by electropositive atoms such as [Sn@Cu12@Sn20]12−,[20] as soluble ions [As@Ni12@As20]3−[13] or as ligand-stabilized molecules such as [Mo(ZnCH3)9(ZnCp*)3].[22]
References
[edit]- ^ Umena, Yasufumi; Kawakami, Keisuke; Shen, Jian-Ren; Kamiya, Nobuo (May 2011). "Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å" (PDF). Nature. 473 (7345): 55–60. Bibcode:2011Natur.473...55U. doi:10.1038/nature09913. PMID 21499260. S2CID 205224374.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ D. Seyferth (1976). Chemistry of Carbon-Functional Alkylidynetricobalt Nonacarbonyl Cluster Complexes. Advances in Organometallic Chemistry. Vol. 14. pp. 97–144. doi:10.1016/s0065-3055(08)60650-4. ISBN 9780120311149.
- ^ A. Bino; M. Ardon; I. Maor; M. Kaftory; Z. Dori (1976). "[Mo3(OAc)6(CH3CH2O)2(H2O)3]2+ and Other New Products of the Reaction between Molybdenum Hexacarbonyl and Acetic Acid". J. Am. Chem. Soc. 98 (22): 7093–7095. doi:10.1021/ja00438a067.
- ^ A. Bino; F. A. Cotton; Z. Dori (1981). "A New Aqueous Chemistry of Organometallic, Trinuclear Cluster Compounds of Molybdenum". J. Am. Chem. Soc. 103: 243–244. doi:10.1021/ja00391a068.
- ^ F. A. Cotton; Z. Dori; M. Kapon; D. O. Marler; G. M. Reisner; W. Schwotzer; M. Shaia (1985). "The First Alkylidyne-Capped Tritungsten(IV) Cluster Compounds: Preparation, Structure, and Properties of [W3O(CCH3)(O2CCH3)6(H2O)3]Br2*2H2O". Inorg. Chem. 24: 4381–4384. doi:10.1021/ic00219a036.
- ^ Arndt Simon "Metal clusters inside out" Phil. Trans. R. Soc. A 2010 vol. 368, 1285-1299. doi:10.1098/rsta.2009.0271
- ^ Koknat, F. W.; Marko, D. J. "Tetradecachlorohexatantalum Octahydrate, Ta6Cl14.8H2O" Inorganic Syntheses, 2004, volume 34, pp. 187-191. ISBN 0-471-64750-0. (describes Na4Ta6Cl18)
- ^ Jacobson, Robert A.; Thaxton, Charles B. (1971). "The Crystal Structure of H2(Ta6Cl18)(H2O)6". Inorganic Chemistry. 10 (7): 1460–1463. doi:10.1021/ic50101a029.
- ^ "Metal Clusters in Chemistry" P. Braunstein, L. A. Oro, P. R. Raithby, eds Wiley-VCH, Weinheim, 1999. ISBN 3-527-29549-6.
- ^ Lee, Sonny C.; Lo, Wayne; Holm, R. H. (2014). "Developments in the Biomimetic Chemistry of Cubane-Type and Higher Nuclearity Iron–Sulfur Clusters". Chemical Reviews. 114 (7): 3579–3600. doi:10.1021/cr4004067. PMC 3982595. PMID 24410527.
- ^ S. Scharfe; F. Kraus; S. Stegmaier; A. Schier; T. F. Fässler (2011). "Homoatomic Zintl Ions, Cage Compounds, and Intermetalloid Clusters of Group 14 and Group 15 Elements". Angewandte Chemie International Edition. 50 (16): 3630–3670. doi:10.1002/anie.201001630. PMID 21455921.
- ^ a b c Zintl Ions: Principles and Recent Developments, Book Series: Structure and Bonding. T. F. Fässler (Ed.), Volume 140, Springer, Heidelberg, 2011 doi:10.1007/978-3-642-21181-2
- ^ A. Spiekermann; S. D. Hoffmann; T. F. Fässler (2006). "The Zintl Ion [Pb10]2−: A Rare Example of a Homoatomic closo Cluster". Angewandte Chemie International Edition. 45 (21): 3459–3462. doi:10.1002/anie.200503916. PMID 16622888.
- ^ itself made by heating elemental potassium and lead at 350°C
- ^ Tin particles are generated as K+Sn122− by laser evaporation from solid tin containing 15% potassium and isolated by mass spectrometer before analysis
- ^ Li-Feng Cui; Xin Huang; Lei-Ming Wang; Dmitry Yu. Zubarev; Alexander I. Boldyrev; Jun Li; Lai-Sheng Wang (2006). "Sn122−: Stannaspherene". J. Am. Chem. Soc. 128 (26): 8390–8391. doi:10.1021/ja062052f. PMID 16802791.
- ^ J.-Q. Wang; S. Stegmaier; B. Wahl; T. F. Fässler (2010). "Step by Step Synthesis of the Endohedral Stannaspherene [Ir@Sn12]3− via the Capped Cluster Anion [Sn9Ir(COD)]3−". Chem. Eur. J. 16 (6): 3532–3552. doi:10.1002/chem.200902815. PMID 20077544.
- ^ A. Schnepf; H. Schnöckel (2002). "Metalloid aluminum and gallium clusters: element modifications on the molecular scale?". Angewandte Chemie International Edition. 114 (19): 1793–1798. doi:10.1002/1521-3773(20021004)41:19<3532::AID-ANIE3532>3.0.CO;2-4. PMID 12370894.
- ^ a b S. Stegmaier; T. F. Fässler (2011). "A Bronze Matryoshka – The Discrete Intermetalloid Cluster [Sn@Cu12@Sn20]12− in the Ternary Phases A12Cu12Sn21 (A = Na, K)". J. Am. Chem. Soc. 133 (49): 19758–19768. doi:10.1021/ja205934p. PMID 21961732.
- ^ T. F. Fässler; S. D. Hoffmann (2004). "Endohedral Zintl Ions: Intermetalloid Clusters". Angewandte Chemie International Edition. 116 (46): 6400–6406. doi:10.1002/anie.200460427. PMID 15505810.
- ^ R. A. Fischer; et al. (2008). "Twelve One-Electron Ligands Coordinating One Metal Center: Structure and Bonding of [Mo(ZnCH3)9(ZnCp*)3]". Angewandte Chemie International Edition. 47 (47): 9150–9154. doi:10.1002/anie.200802811. PMID 18846517.
