User:Mr. Ibrahem/Amodiaquine
{{#unlinkedwikibase:id=Q239569}}
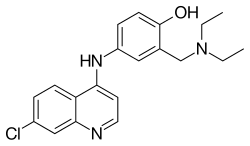 | |
| Clinical data | |
|---|---|
| Trade names | Amdaquine, Amobin, others[1] |
| AHFS/Drugs.com | International Drug Names |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C20H22ClN3O |
| Molar mass | 355.861 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Amodiaquine (ADQ) is a medication used to treat malaria, including Plasmodium falciparum malaria when uncomplicated.[4][5] It is recommended to be given with artesunate to reduce the risk of resistance.[4] Due to the risk of rare but serious side effects, it is not generally recommended to prevent malaria.[4] Though, the WHO in 2013 recommended use for seasonal preventive in children at high risk in combination with sulfadoxine and pyrimethamine.[6]
The side effects of amodiaquine are generally minor to moderate and are similar to those of chloroquine.[5] Rarely liver problems or low blood cell levels may occur.[4] When taken in excess headaches, trouble seeing, seizures, and cardiac arrest may occur.[4] While not extensively studied as of 2007, it appeared to be safe in pregnancy.[7] Amodiaquine is a 4-aminoquinoline compound related to chloroquine.[4]
Amodiaquine was first made in 1948.[8] It is on the World Health Organization's List of Essential Medicines.[9] The wholesale cost is about US$0.01 per dose as of 2014.[10] While not available in the United States,[11] it is widely available in Africa.[4][12]
References[edit]
- ^ "Amodiaquine". drugs.com. Archived from the original on 27 November 2016. Retrieved 27 November 2016.
- ^ "AMODIAQUINE = AQ oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 24 January 2021. Retrieved 25 August 2020.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 25 July 2020. Retrieved 21 September 2020.
- ^ a b c d e f g Nair, A; Abrahamsson, B; Barends, DM; Groot, DW; Kopp, S; Polli, JE; Shah, VP; Dressman, JB (December 2012). "Biowaiver monographs for immediate release solid oral dosage forms: amodiaquine hydrochloride". Journal of Pharmaceutical Sciences. 101 (12): 4390–401. doi:10.1002/jps.23312. PMID 22949374.
- ^ a b Olliaro, P; Mussano, P (2003). "Amodiaquine for treating malaria". The Cochrane Database of Systematic Reviews (2): CD000016. doi:10.1002/14651858.CD000016. PMC 6532704. PMID 12804382.
- ^ Seasonal malaria chemoprevention with sulfadoxine–pyrimethamine plus amodiaquine in children: a field guide (PDF). Geneva: The World Health Organization. August 2013. ISBN 978 92 4 150473 7. Archived (PDF) from the original on 2020-07-26. Retrieved 2020-01-14.
- ^ Tagbor, HK; Chandramohan, D; Greenwood, B (November 2007). "The safety of amodiaquine use in pregnant women". Expert Opinion on Drug Safety. 6 (6): 631–5. doi:10.1517/14740338.6.6.631. PMID 17967151.
- ^ Profiles of Drug Substances, Excipients and Related Methodology. Academic Press. 1992. p. 45. ISBN 9780080861166. Archived from the original on 2017-09-08.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Amodiaquine". International Drug Price Indicator Guide. Archived from the original on 29 March 2019. Retrieved 4 December 2015.
- ^ "Amodiaquine". Livertox. Archived from the original on 27 November 2016. Retrieved 27 November 2016.
- ^ Centers for Disease Control, (CDC). (12 April 1985). "Revised recommendations for preventing malaria in travelers to areas with chloroquine-resistant Plasmodium falciparum". MMWR. Morbidity and Mortality Weekly Report. 34 (14): 185–90, 195. PMID 3156271.
