User:Mr. Ibrahem/Paliperidone
 | |
| Clinical data | |
|---|---|
| Trade names | Invega, Xeplion, Trevicta, others |
| Other names | 9-hydroxyrisperidone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607005 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (OROS tablets), intramuscular |
| Drug class | Atypical antipsychotic[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 28% (by mouth) |
| Elimination half-life | 23 hours (by mouth) |
| Excretion | 1% unchanged in urine 18% unchanged in feces |
| Identifiers | |
| |
| Chemical and physical data | |
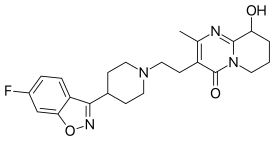
| Formula | C23H27FN4O3 |
| Molar mass | 426.492 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Paliperidone, sold under the trade name Invega among others, is an atypical antipsychotic used to treat schizophrenia and schizoaffective disorder.[1] It may be taken by mouth or injected into a muscle.[5] When the unpublished literature is included, benefits are lower and harms are higher than when only the published literature is looked at.[6]
Common side effects include headache, trouble sleepiness, parkinsonism, involuntary muscle movements, dizziness, agitation, depression, weight gain, nausea, dry mouth, QT prolongation, and tiredness.[1] Other side effects may include neuroleptic malignant syndrome, tardive dyskinesia, diabetes, and high prolactin.[5] In older people with dementia, use increases the risk of death.[5] Use during pregnancy may have negative effects on the baby.[7] Benefits are believed to be due to effects on dopamine and serotonin.[5]
Paliperidone was approved for medical use in the United States in 2006 and Europe in 2007.[5][1] It is on the World Health Organization's List of Essential Medicines.[8] In the United Kingdom the typical dose by mouth cost the NHS about £105 per month as of 2021.[9] This amount in the United States costs about 400 USD.[10]
References[edit]
- ^ a b c d e f "Invega". Archived from the original on 9 January 2021. Retrieved 25 October 2021.
- ^ "Invega- paliperidone tablet, extended release". DailyMed. Archived from the original on 24 January 2016. Retrieved 19 August 2020.
- ^ "Invega Sustenna- paliperidone palmitate injection". DailyMed. 31 January 2019. Archived from the original on 13 May 2021. Retrieved 19 August 2020.
- ^ "Invega Trinza- paliperidone palmitate injection, suspension, extended release". DailyMed. 31 January 2019. Archived from the original on 17 June 2021. Retrieved 19 August 2020.
- ^ a b c d e f "Paliperidone Monograph for Professionals". Drugs.com. Archived from the original on 25 February 2021. Retrieved 25 October 2021.
- ^ Hodkinson, A; Heneghan, C; Mahtani, KR; Kontopantelis, E; Panagioti, M (25 August 2021). "Benefits and harms of Risperidone and Paliperidone for treatment of patients with schizophrenia or bipolar disorder: a meta-analysis involving individual participant data and clinical study reports". BMC medicine. 19 (1): 195. doi:10.1186/s12916-021-02062-w. PMID 34429113.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Paliperidone Use During Pregnancy". Drugs.com. Archived from the original on 4 December 2020. Retrieved 25 October 2021.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 422. ISBN 978-0857114105.
- ^ "Invega Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 25 January 2021. Retrieved 25 October 2021.
